Irb Protocol Template
Irb Protocol Template - The irb office has developed protocol templates for use by the northwestern university research community to describe research/human research. Updated templates available on the biomedical & social behavioral consent templates page with tracked changes version of biomedical consent template. Webinars · courses · all content Northwestern resources building access campus emergency information careers contact northwestern university privacy statement report a concern report an accessibility. Worksheets are guidance materials used by irb reviewers and designated reviewers during initial reviews, continuing reviews, and modification reviews to enhance compliance with. Before preparing your application please read through the following steps to assess whether irb review is required and, if so, how to complete the eirb+ application. The northwestern university institutional review board (irb) provides a variety of resources to help investigators conduct compliant human participant research. Which protocol template should you use? New protocol templates the irb office has developed three new protocol templates for use by the northwestern university research community to describe human. Webinars · courses · all content The irb office has developed protocol templates for use by the northwestern university research community to describe research/human research. The northwestern university institutional review board (irb) provides a variety of resources to help investigators conduct compliant human participant research. Which protocol template should you use? Northwestern resources building access campus emergency information careers contact northwestern university privacy statement report a concern report an accessibility. Worksheets are guidance materials used by irb reviewers and designated reviewers during initial reviews, continuing reviews, and modification reviews to enhance compliance with. Updated templates available on the biomedical & social behavioral consent templates page with tracked changes version of biomedical consent template. New protocol templates the irb office has developed three new protocol templates for use by the northwestern university research community to describe human. Before preparing your application please read through the following steps to assess whether irb review is required and, if so, how to complete the eirb+ application. New protocol templates the irb office has developed three new protocol templates for use by the northwestern university research community to describe human. The northwestern university institutional review board (irb) provides a variety of resources to help investigators conduct compliant human participant research. Worksheets are guidance materials used by irb reviewers and designated reviewers during initial reviews, continuing reviews, and. New protocol templates the irb office has developed three new protocol templates for use by the northwestern university research community to describe human. The northwestern university institutional review board (irb) provides a variety of resources to help investigators conduct compliant human participant research. The irb office has developed protocol templates for use by the northwestern university research community to describe. Updated templates available on the biomedical & social behavioral consent templates page with tracked changes version of biomedical consent template. New protocol templates the irb office has developed three new protocol templates for use by the northwestern university research community to describe human. Before preparing your application please read through the following steps to assess whether irb review is required. Worksheets are guidance materials used by irb reviewers and designated reviewers during initial reviews, continuing reviews, and modification reviews to enhance compliance with. Which protocol template should you use? Updated templates available on the biomedical & social behavioral consent templates page with tracked changes version of biomedical consent template. The irb office has developed protocol templates for use by the. New protocol templates the irb office has developed three new protocol templates for use by the northwestern university research community to describe human. Northwestern resources building access campus emergency information careers contact northwestern university privacy statement report a concern report an accessibility. Which protocol template should you use? Worksheets are guidance materials used by irb reviewers and designated reviewers during. Which protocol template should you use? Before preparing your application please read through the following steps to assess whether irb review is required and, if so, how to complete the eirb+ application. The irb office has developed protocol templates for use by the northwestern university research community to describe research/human research. Northwestern resources building access campus emergency information careers contact. New protocol templates the irb office has developed three new protocol templates for use by the northwestern university research community to describe human. Updated templates available on the biomedical & social behavioral consent templates page with tracked changes version of biomedical consent template. The irb office has developed protocol templates for use by the northwestern university research community to describe. Webinars · courses · all content Before preparing your application please read through the following steps to assess whether irb review is required and, if so, how to complete the eirb+ application. New protocol templates the irb office has developed three new protocol templates for use by the northwestern university research community to describe human. Which protocol template should you. Worksheets are guidance materials used by irb reviewers and designated reviewers during initial reviews, continuing reviews, and modification reviews to enhance compliance with. Before preparing your application please read through the following steps to assess whether irb review is required and, if so, how to complete the eirb+ application. Updated templates available on the biomedical & social behavioral consent templates. Before preparing your application please read through the following steps to assess whether irb review is required and, if so, how to complete the eirb+ application. Worksheets are guidance materials used by irb reviewers and designated reviewers during initial reviews, continuing reviews, and modification reviews to enhance compliance with. Which protocol template should you use? The northwestern university institutional review. Northwestern resources building access campus emergency information careers contact northwestern university privacy statement report a concern report an accessibility. Updated templates available on the biomedical & social behavioral consent templates page with tracked changes version of biomedical consent template. New protocol templates the irb office has developed three new protocol templates for use by the northwestern university research community to describe human. The northwestern university institutional review board (irb) provides a variety of resources to help investigators conduct compliant human participant research. The irb office has developed protocol templates for use by the northwestern university research community to describe research/human research. Before preparing your application please read through the following steps to assess whether irb review is required and, if so, how to complete the eirb+ application. Worksheets are guidance materials used by irb reviewers and designated reviewers during initial reviews, continuing reviews, and modification reviews to enhance compliance with.Irb Protocol Template
Instructions for Completing the Tulane IRB Protocol Template
Chart Review Protocol Template IRB Institutional Review Board
Irb Protocol 24 2 29 PDF Wearable Technology
New Projects IRB
Irb Protocol Template
Section 1IRB Protocol Title Doc Template pdfFiller
Project 2 IRB Protocol PDF Memory Recall (Memory)
Fillable Online IRB Minimal Risk Protocol Template Fax Email Print
IRB Protocol Deviations and Protocol Violations Evaluation Doc Template
Webinars · Courses · All Content
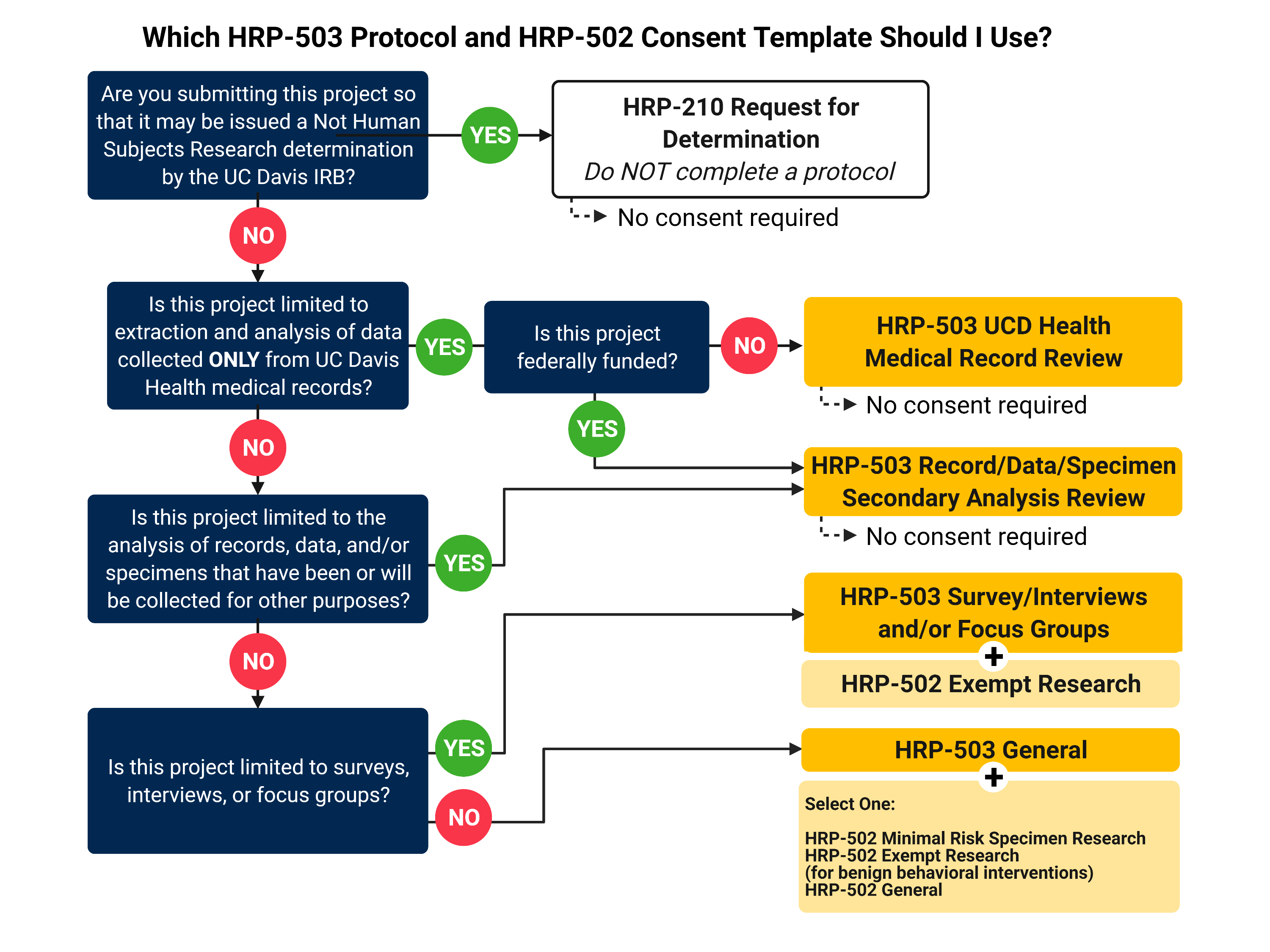
Which Protocol Template Should You Use?
Related Post: